Sandoz Medical Device
Human factors and device development
Human factors and device development
Sandoz was developing a new dry powder inhaler with a dose-by-dose counter mechanism. The tolerance chains for the counter components were large relative to the viewing window — meaning at extremes of manufacturing variation, digits could misalign enough to be partially masked. Before regulatory submission, they needed hard evidence that the counter remained readable across every tolerance condition, including worst-case misalignment. The question wasn't whether the device worked. It was whether users could trust what they read.
Working directly with Rebus Medical and Sandoz's lead industrial engineer, I took ownership of the full mechanical methodology for the study. Each of 24 prototype inhalers needed its dose counter locked at a precise angular offset - some within expected manufacturing tolerance, some at the extreme ends, some deliberately beyond. I designed custom positional offset components with precision dowel holes to fix each counter wheel at exact angles, machined in collaboration with our in-house CNC machinist. Before any device entered the study, every offset was validated using a Cognex In-Sight vision system with adapted C++ inspection code, confirming each angle matched its specification. This process produced submission-grade data - it had to, because the output would go directly into a regulatory evidence package.
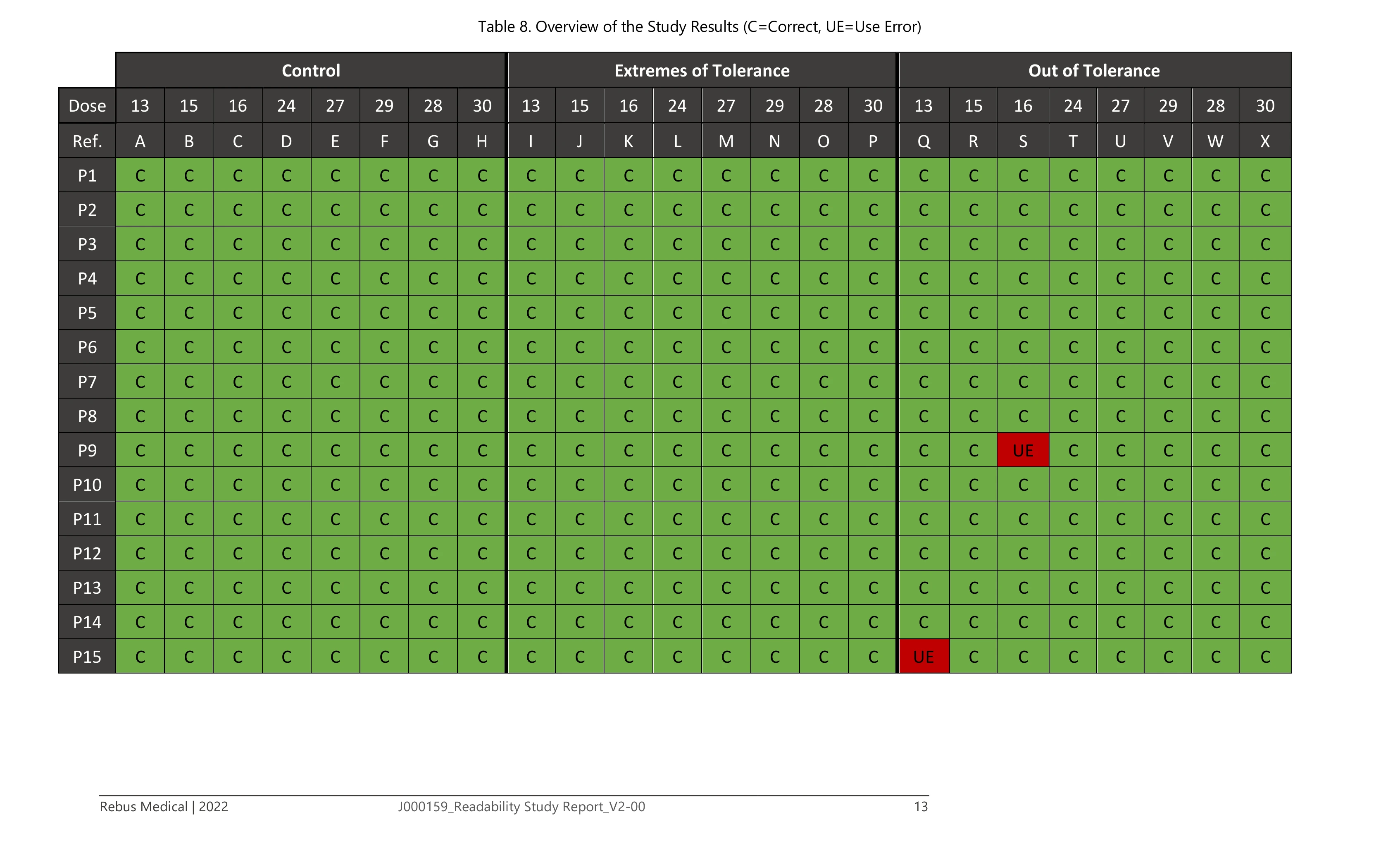
The study used three groups of eight devices each. A control group with digits presented nominally. An extremes-of-tolerance group with the tens digit offset to approximately 6.6° high or 5° low, and the units digit to approximately 3°. And an outside-tolerance group pushed beyond that - tens at approximately 10° and units at approximately 5.5°. Dose values were chosen specifically from digit pairs that are commonly confused: 1/7, 5/6, 2/7, 8/9. 15 patients with COPD or asthma - all current Ellipta inhaler users - were each presented with all 24 devices in randomised order and asked to read the dose counter aloud. Sessions were recorded through two cameras and observed through a one-way mirror.
The study returned 358 correct readings from 360 total responses. Both errors came from the outside-tolerance group - conditions that no marketed device will ever reach - and both participants self-corrected immediately on a second look. Across the control and extremes-of-tolerance groups, accuracy was 100% with no use difficulties recorded. What made those results stand up was the mechanical preparation: precision offset jigs, independent Cognex vision verification of every device angle before it entered the study room, and documentation structured for regulatory submission from the outset. The data gave Sandoz what they needed: clear evidence of readability across all achievable manufacturing conditions. The conclusion was direct: 100% accuracy within tolerance, 98% outside - and marketed devices will never reach that condition.
The placement covered significantly more ground than what's shown here - much of it under NDA. What follows is the second project I can speak to in full.
Autoinjector Characterisation
and Mechanism Design
I designed and built a modular test rig to characterise over twelve on-market biosimilar autoinjectors across five force and performance parameters: actuation force, cap removal force, injection time, drug substrate volume, and lockout break force. The rig was built around an SLA-printed modular fixture system, each fixture reverse engineered from physical autoinjectors into CAD and printed to hold each device safely and repeatably. That modular approach meant every test could be run on a single Mecmesin MultiTest frame, controlled through a custom automated script that replicated the full autoinjector use procedure without manual intervention. All testing was conducted in a clean bio lab under sharps safety and drug handling regulations. The output was a validated system requirements specification, formed directly from measured on-market device performance. Because we were developing a biosimilar, that data served as regulatory evidence of suitability, letting us move significantly faster than a first-in-class device programme would allow.
Rowan and I then designed the autoinjector from scratch against our own specification. The constraints were real: single-use lockout reliability, a spring-actuated delivery system, and a patent landscape where locking mechanisms and drug delivery geometry were heavily protected. Every geometric decision had a legal dimension. We mapped the relevant patents first, then designed around them, developing a novel latch system that achieved lockout without infringing on any existing protected mechanism.
The entire design went from specification to a working 3D printed prototype. That prototype locks out correctly after injection, delivers drug volume within specification, and the latch engages under spring load every time. Fourteen components total, down from over twenty in comparable on-market devices. The novel latch entered early-stage patenting at the close of the placement.
Later in the year, Sandoz's Global Head of Device Development came to see the work. Rowan and I presented directly to her. That meeting wasn't in the original plan. The mechanism earned it.
Total device readings across 15 participants. 100% accuracy within manufacturing tolerance. Both use errors came from the outside-tolerance group - a condition no marketed device will reach - and both self-corrected on a second look. Submitted as regulatory evidence to Sandoz.
Components in the prototype, down from over 20 in comparable on-market devices. Characterisation data from twelve biosimilar autoinjectors became the validated system requirements specification. A novel latch and lockout mechanism entered early-stage patenting at the close of the placement.

Two placement students were handed a blank page and told to build an autoinjector platform from scratch. Six weeks to a working prototype. By the end of the year the latch mechanism was entering patenting, the readability study data was in a regulatory submission, and we'd had an unscheduled design review with the Global Head of Device Development because the work made it worth her time. The trust Sandoz gave us to explore and own real problems gave me more practical design engineering experience in one year than my entire degree had.
This placement shows I can operate inside a regulated development environment, take ownership of engineering work that feeds directly into regulatory submissions, and apply rigorous mechanical thinking to problems where getting it wrong has real consequences. It also shows I can move fast when the brief demands it - characterising twelve devices, specifying a system, and prototyping a patentable mechanism inside twelve months.